Observe the decay of airborne radionuclides with a Geiger counter and computer. (OK, it's not new since we've been doing the experiment for 20 years...we just neglected adding it to our list.)
What it shows:
Filtering dust particles from the air is a standard procedure for monitoring radiation levels. The experiment uses a charged balloon to extract the dust (and any radionuclides residing on it) from the air. The radiation level of the balloon is monitored by a Geiger counter and the half-life of the decay products is observed.
How it works:
All of the heavy elements (Z>83) found in nature are radioactive and decay by alpha or beta emission. Furthermore, all the naturally occurring heavy radionuclides belong to one of three series: (1) 238U-Radium, (2) 235U-Actinium, and (3) 232Thorium. All three series contain one gaseous member (an isotope of Rn) and end in a stable isotope of Pb.
The radium series starts with 238U. Uranium and its first five daughters are solids that remain in the soil, but the fifth daughter 226Ra decays into 222Rn. This daughter, called radon, is a noble gas, not bound chemically in the material where its parents resided. The half-life of 222Rn (3.82 d) is long enough for much of the gas to work its way out into the atmosphere. Radon is also generated in the other two series. However, these isotopes of radon are of lesser radiological importance. The thorium series generates 220Rn, which is also called thoron. 220Rn has a half-life of 56 s and therefore has a much greater chance to decay before becoming airborne. The actinium series produces 219Rn, also called actinon, after several transformations from the relatively rare original 235U nuclide. Its half-life is only 4 s, and its contribution to airborne radon is insignificant. Thus we will consider only radon from the radium series. The decay chain of radon is as follows:1

The technique of using a balloon for extracting radioactive substances from the air was brought to our attention by T.A. Walkiewicz.2 The daughter products of radon become attached to positive charged aerosol particles. These particles are readily attracted to a negative charged object (a balloon, in this case), thereby building up a radioactive source with a compound half-life of about ?? minutes. Read on to learn why we state the half-life half-life as ?? minutes.
Since we do not know in what proportion the daughter nuclides of radon have been collected on the balloon, it is not at all clear what half-life has been measured. However, we can infer the following. First, the half-life of Po-218 is short enough that its activity will be greatly reduced after several minutes and can be completely ignored for times longer than that. For example, in 21 minutes (7 half-lives) the amount of Po-218 will have dropped to 1/128 of the original amount. The effective half-life of the radioactive balloon is then governed mainly by the activities of Pb-214 and Bi-214, which have half-lives of 27 and 20 minutes, respectively. If the data are teated as due to a single, effective half-life, the value determined will depend on both the relative amounts of these isotopes that were collected in the first place, as well as their decay. Furthermore, although the balloon emits alpha particles and gamma rays, the majority of activity detected by the Geiger counter is beta particles from Pb-214 and Bi-214. Gamma rays have a much lower detection efficiency than do beta particles for a Geiger counter, and many of the alpha particles from Po-218 are absorbed by the balloon, the intervening air, and the Geiger tube wall. Thus, you may find that the plot of the radioactive decay actually rises (CPM goes up) in the first 20 minutes or so, levels off, and then decreases. This seemingly strange behavior can be attributed to the fact that the radiation monitor is not as sensitive to the Po-218 decay but, as the amount of Pb-214 builds up (due to the Po-218 decay), the count rate goes up because the monitor is sensitive to the decay of Pb-214. Detector "dead time" has not been measured and may also have an effect. In conclusion, the effective half-life measurement is not at all a "clean" measurement. Having said that, the following is a screen shot of an actual 1.5 hour-long run in lecture hall A.
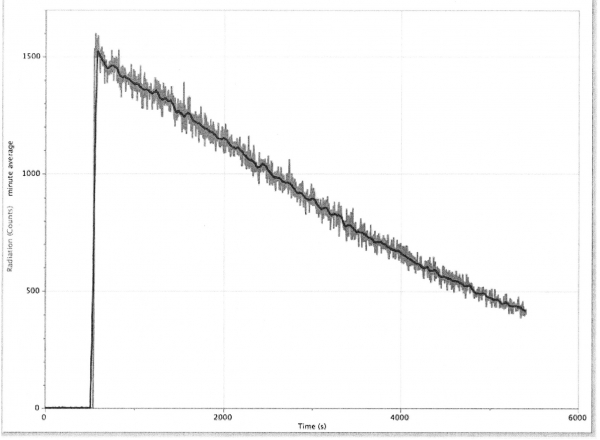
The binning on the horizontal scale is 3 seconds, so the 1500 counts on the vertical scale corresponds to an astonishing 30,000 cnts/min. The background rate was about 65 cnts/min, so the initial balloon activity was over 460 times the background! The count rate dropped to 1/2 its original value after about 3000 seconds, or 50 minutes. Clearly, the measured decay is not exponential in character in the first 1.5 hours, so complicated things are going on.
The next screen shot is of a 12 hour run to see if the measured decay looks more exponential in the long run (the binning here is 5 seconds).
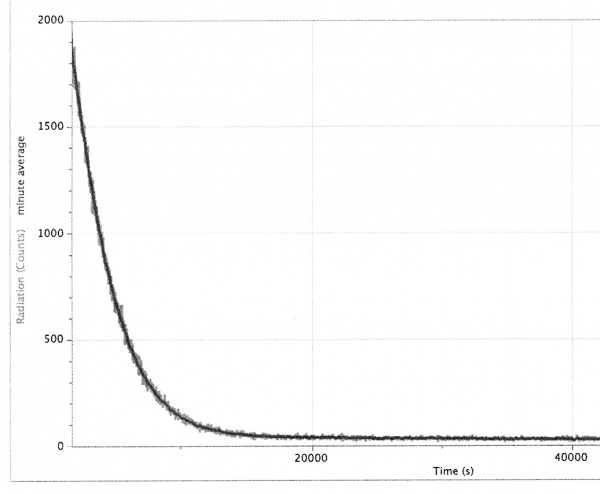
For some reason this run was closer to what one might expect: starting around 45 minutes into the run, the half-life appears to be about 38 minutes. It drops down to around 35 minutes in the hours after that, which is still significantly greater than what one would expect from Pb-214 and Bi-214, which have half-lives of 27 and 20 minutes, respectively. After 17 hours, the activity was still about 150 cpm, or about twice background.
To help decipher the curiously long half-life observed, the experiment was repeated and the balloon was placed in a NaI scintillation spectrometer. Five very strong gamma peaks were observed. Two of them were identified as gammas associated with Pb-214 (353 keV) and Bi-214 (609 keV) decay, both daughters of radon. On the other hand, a strong 239 keV peak was most likely due to the presence of Pb-212, a daughter of thoron. Pb-212 decays into Bi-212 by β emission (0.346 MeV) and has a half-life of 10.6 hr (details are given in the Thoron Decay demonstration below). Apparently, even though thoron has a half-life of only 56 seconds, enough is getting out of the ground so that its progeny is also collected by the balloon. One further note, the 727 keV gamma associated with the decay of Bi-212 (half-life = 60.6 min) was not observed. Only 7% of the decays give off this gamma and consequently its signature was too weak to appear in the background.
Setting it up:
Suspend a 12" diameter balloon in such a way that it is relatively far away from everything around it, as it is easily attracted to anything within reach. Charge the balloon by rubbing it with rabbit fur and leave it undisturbed for approximately 30 minutes. The balloon is then removed from the string and carefully deflated by piercing a hole in the inflation end. It is then flattened out on the table and a Geiger-Mueller counter3placed on top. The output of the counter is connected to an iMac via a Vernier LabQuest interface. Logger Pro software (radon_daughter.cmbl) displays the incoming counts vs time, much like a multi-channel scaler. The initial count rate is surprisingly high — over 460 times the background rate. We typically take data for 1 hour or more. Thus, it's best to charge the balloon 1/2 hour before class if you wish to see one or two half lives in the decay.
Comments:
Airborne radon itself poses little health hazard. As an inert gas, inhaled radon is not retained in significant amounts by the body — it does not stick to the walls of the respiratory tract or deep in the lungs. The potential health hazard arises when radon in the air decays, producing non-gaseous radioactive daughters — the daughters are reactive heavy metals that quickly become attached to things in the room such as furniture or, in the case of room air, smoke, water vapor, or dust, often collectively called aerosols. In this form these radioactive particles may be drawn into the respiratory tract or deep into the lungs. In this situation, when an alpha particle is emitted inside the lung, it deposits all of its energy locally within a small thickness of adjacent tissue. A beta particle emitted deposits its energy over a much larger distance of about 4 mm.5
References:
1. F.W. Walker, D.G. Miller and F. Feiner, Chart of the Nuclides, (General Electric Co., San Jose CA, 1984).
2. T.A. Walkiewicz, "The Hot Balloon (Not Air)," The Phys Teacher 33, 344-345 (Sept 1995). An earlier article by J.C. Cowie, Jr. and T.A. Walkiewicz, "Radioactiveball," TPT 30, 16-17 (Jan 1992) measured the radioactivity of a handball, after it had been in play for 50 minutes.
3. The counter is the Radalert Inspector model made by International Medcom. It has a relatively large window (1.75" dia) with a density of 1.5-2.0 mg/cm2, ideal for this application. The dual miniature output jack drives CMOS or TTL devices, sending counts to computer or data logger.
4. According to the spec sheet, the Inspector is only about 36% efficient in detecting alphas whereas it is 65% efficient in detecting the 1 MeV betas (from Bi-214), and 80% efficient in detecting the 3 MeV betas (from Pb-214).
5. See M. Lafavore, Radon, (Rodale, Emmaus PA, 1987) and J.E. Turner, Atoms, Radiation, and Radiation Protection, 2nd ed (Wiley & Sons, NY, 1995) for more information..