Nitrogen dioxide is produced by an electric discharge in air and, when sprayed with a water mist, produces acid rain.
What it shows:
Some of the most irritating and dangerous pollutants in our atmosphere are gases such as sulfur dioxide and nitrogen dioxide. Nitrogen dioxide is a deep orange-red gas that, together with smokelike particles, is responsible for the color of smog. In this demonstration, nitrogen dioxide is produced by an electric discharge in air and, when sprayed with a water mist, produces acid rain.
How it works:
A large glass flask is filled with air at atmospheric pressure and placed in front of a photographic light box for back-light illumination. High voltage, applied to two electrodes inside the flask, produces an electrical discharge through the air. Within one minute, the air inside the flask turns noticeably orange-brown. With time, the color of the air becomes increasingly more saturated and, after about five minutes, has that distinctive dark smog look. The light box provides an excellent (white) color reference as well as contrast.
If desired, the flask can be opened (after the high voltage is turned off) and a fine mist of water sprayed into the bottle produces a yellow-brown cloud. As more and more water is sprayed in, the precipitation begins to collect on the bottom of the flask. When enough has accumulated, the water can be dumped out into a small beaker for analysis. Indicator strips show a strong acidity with pH=1 or 2. Alternatively, a few ml of blue Bromcresol Green added to the clear (acidic) water turns the mixture yellow!
The nitrogen molecule is quite inert because its atoms are bound together by a triple bond: N≡N. Once these bonds are broken, nitrogen is highly reactive and forms numerous compounds. Nitric oxide (NO), a colorless and slightly toxic gas, is one such product in the reaction of N2 and O2 in the atmosphere:![]()
The equilibrium constant for this reaction is only 6.4 × 10-16 at 25°C, so very little NO forms at room temperature. 1 However, the equilibrium constant increases by a huge factor with increasing temperature to a value of 0.013 at 3000K. 2 Nitric oxide forms brown fumes of nitrogen dioxide (NO2) instantaneously in the presence of air by
2NO + O2 → 2NO2
Unlike nitrous oxide (N2O) and nitric oxide, nitrogen dioxide is a highly toxic gas with a choking odor. Both nitric oxide and nitrogen dioxide are formed by the combination of nitrogen and oxygen in the combustion chambers and hot exhausts of engines, and they are collectively known as NOx. In this demonstration, the high temperatures needed to break the nitrogen triple bond are present in the hot plasma of the electrical discharge through the air in the flask. This is similar to lightning through the atmosphere which too produces an appreciable amount of nitric oxide. Upon cooling and mixing with more oxygen the brown dioxide is produced.
Nitrogen dioxide is an acidic oxide; it reacts rapidly with cold water to form both nitrous acid and nitric acid:
2NO2 + H2O → HNO2 + HNO3
as well as nitric acid and nitric oxide:
3NO2 + H2O → 2H+ + 2NO3- + NO
wherein
2H+ + 2NO3- → 2HNO3
These acids are formed in the atmosphere when nitrogen dioxide dissolves in water and precipitate as one component of what is generically called acid rain.
Setting it up:
The 12 liter (28 cm dia.) flask is sealed with a rubber stopper that serves as a holder for two long, copper electrodes configured in a V-shape. 15,000 VAC (30 mA) is supplied to the electrodes by a luminous tube transformer and produces a Jacob's Ladder plasma arc inside the flask. Obvious precautions should be observed in using this lethal high voltage. Furthermore, care should be taken not to breathe the highly toxic gases inside the flask. In addition to the nitrogen oxides, ozone (O3), a pungent toxic gas is also produced by the electric discharge. Thus the flask should be vented in a fume hood or outdoors after the demonstration.
Concerning the acid indicator, the color range of Bromcresol Green is yellow to blue with a pH range of 4.0 to 5.6 respectively. Bromphenol Blue could also be used with a color range of yellow to blueviolet and a corresponding pH range of 3.0 to 4.6. The indicator can be poured into a medium-size petri dish and placed on an OHP so that the entire class can see. Likewise, the highly acidic water from the smog flask can also be placed on the OHP to show that it's clear. Then pour the clear liquid into the blue indicator and the class can observe the dramatic change to a yellow.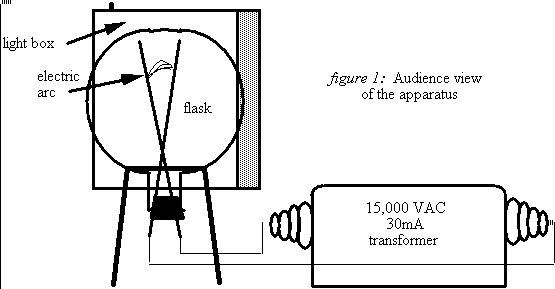
As noted above, color changes in the flask are noticeable after only a minute of operation. By the end of five minutes the NO2 concentration is high enough so that 40% of the transmitted light is absorbed; NO2 production levels off after 10 to 12 minutes at which point 68% of the light is absorbed and the flask appears quite saturated in color. The acid rain experiment can be performed after five minutes of operation.
Comments:
Thanks to Prof. Owen Gingerich for suggesting we pursue the development of a "smog demonstration" in the first place. This is a marvelous demonstration: it's pedagogically simple in its presentation, dramatic, and easy to set up. The Jacob's ladder, a necessary ingredient, adds that showmanship flair. The results are unambiguous.
Going further:
Many of the modern chemistry textbooks discuss this subject matter in greater detail. 3 This demonstration experiment can also be used as an introduction to photochemical smog. Students are usually familiar with the fact that nitrogen dioxide builds up before dawn and in heavy traffic. As the sun rises, its ultraviolet radiation begins to stimulate chemical reaction (photodissociation) and ozone (O3) is produced. 4 NO2 levels decline while O3levels reach their peak shortly after noon:
NO2 + hν → NO + O
Most of the atomic oxygen produced reacts with molecular oxygen in the atmosphere to form ozone. Once some ozone has been formed, it can rapidly oxidize NO to more NO2:
NO + O3 → NO2 + O2
This, of course provides an additional source of NO2 for the formation of more ozone as well as moderating the buildup of ozone. In the late afternoon photochemical reactions slow and ozone is removed from the atmosphere by this reaction with nitric oxide.
In this demonstration, ozone is produced simultaneously with nitric oxide and thus complicates the series of secondary reactions leading to the final equilibrium state in the flask. Indeed, the formation of nitrogen dioxide may very well be tempered by the large quantities of ozone present. Upon close observation of the electric arc and its extinction in the smog flask, one can see a "clear cloud" (in the smog atmosphere) rising. This is most likely evidence of the dissociation of NO2 in the presence of the arc. The clear cloud dissipates rapidly (about 1/2 second) as the NO reverts back to NO2 and gives an indication of what is meant by "instantaneously" in the textbooks.
One can show the molecular absorption spectrum. Being a gas of color, NO2 obviously has absorption resonances in the visible part of the spectrum. This is easily demonstrated by holding the flask in the optical path of a continuous white light spectrum. Many molecular absorption bands are visible throughout the spectrum. The entire blue end of the spectrum (and on into the UV) is absorbed by the gas, as one would guess judging by the color of the gas. This furthermore punctuates the discussion above concerning UV absorption and the consequent dissociation of NO2.
One final comment concerning the production of NO2 and acid rain and the Cretaceous calamity scenario: Researchers have dreamed up a vast range of disastrous effects that would accompany a crash of such mythic proportions. 5 Speeding through the atmosphere, the giant meteorite could have generated so much heat that the NO2 and resulting acidic rainfall would have been as corrosive as battery acid, according to some calculations. 6 The rain would have lowered the pH of the ocean's surface, perhaps making it acidic enough to dissolve the calcium carbonate shells of certain plankton.
1 Raymond Chang, Chemistry, 2nd ed., (Random House, New York, 1984).
2 Terrence J. Swift, Principles of Chemistry - A Models Approach,(Heath & Co., Lexington MA, 1975).
3 Two examples are R.G. Gymer, Chemistry: An Ecological Approach, (Harper & Row, New York, 1973) and J.A. Young, Chemistry, A Human Concern, (Macmillan, New York, 1978). Both authors devote entire chapters to air (and other) pollution and have extensive bibliographies for further reference.
4 P.A. Leighton, Photochemistry of Air Pollution, (Academic, New York, 1961).
5 R. Monastersky, Science News 141, 58 (Jan. 25, 1992). "Closing in on the Killer"
6 That's what astronomer Henry Melosh of the University of Arizona calculates would happen if something six miles across fell from space and smacked into Earth, according to S. Begley in Newsweek, 56 (Nov. 23, 1992) "The Science of Doom"