What it shows:
The very first determination of a half-life for a radioactive decay was made by Rutherford. 1 In a study of the properties of thorium emanation, he found that the intensity of the radiations fell off with time in a geometric progression. That historically important result is reproduced in this demonstration experiment. The gas thoron, or thorium emanation, is an isotope of radon (86Rn220) which decays by α emission and has a half life of 55.6 seconds. 2 Using an emanation electroscope, we observe the radioactive decay of thoron (separated from all its parent substances) by the progressive decrease of ionization in the air around the thoron.
How it works:
We use thorium (in thorium oxide) to generate thoron. The sequence of decays is known as the Thorium-232 series and goes as follows:
| isotope | (alternate name) | particle emitted | half-life |
| 90Th232 | α | 1.40 × 1010 yr | |
| 88Ra228 | (MsTh1) | β | 5.76 yr |
| 89Ac228 | (MsTh2) | β | 6.13 hr |
| 90Th228 | (RdTh) | α | 1.91 yr |
| 88Ra224 | (ThX) | α | 3.66 d |
| 86Rn220 | (Thoron, Tn, Em) | α | 55.6 sec |
| 84Po216 | (ThA) | α | 0.15 sec |
| 82Pb212 | (ThB) | β | 10.6 hr |
| 83Bi212 | (ThC) | β | 60.6 min |
| 84Po212 | (ThC') | α | 3.0 × 10-7 sec |
| 82Pb208 | (ThD, lead) | none | stable end product |
Note that a lesser branch at ThC by α decay to ThC (81Tl208), and then to lead-208 by β decay, has been omitted here.
The apparatus used for measuring the radioactive decay is based on a design which has been in use as far back as the work of Rutherford and Thomson and was built by K.T. Bainbridge and J.C. Street; 3 the amount of radioactivity is measured through its effect on the conduction of electricity by gases. The α particles emitted by the thoron ionize the air inside an electroscope; the ionized air causes the electroscope to discharge. The rate of discharge is directly proportional to the activity of the radioisotope. A simplified drawing of the apparatus is given below. 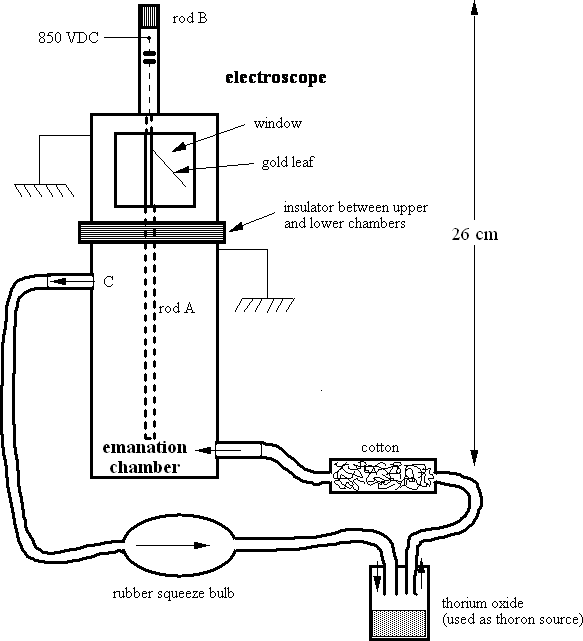
The clean, still air in the upper part of the brass cylinder (the electroscope proper) is completely separated from the emanation chamber beneath it. The leaf of the electroscope in the upper chamber is attached to a conducting rod A, which penetrates into the lower chamber. Thoron gas, released from a preparation of thorium oxide by the radioactive decay of thorium, can be forced into the clean, dry lower chamber by pressing the rubber bulb. The displaced gas leaves through outlet C. The system is closed with respect to the outside, and the gases remain quite stationary after the bulb is released. This is important because we wish to measure the decay of a given amount of thoron after it has been pumped into the lower chamber. By depressing rod B, rod A is contacted and charged to a positive potential of about 850 volts.
The thoron in the lower chamber undergoes radioactive decay with the emission of high energy alpha particles. These alpha particles lose their energy by ionizing the air molecules (the 6.3 MeV alpha has a range of about 4.7 cm in air) which in turn neutralize the positive rod and cause the electroscope leaf to fall. The rate at which the thoron emits alpha rays determines the ionization of air in the lower electroscope chamber and hence, the rate at which charge leaks off the electroscope. The rate of discharge is ascertained by measuring the time required for the leaf to fall a predetermined distance.
The electroscope can be recharged back to the original potential by pushing down rod B to make momentary contact with the stem of rod A. Thus, without adding any additional thoron, the electroscope is repeatedly charged and the "fall time" (the time for the leaf to fall a predetermined distance) recorded. The fall time will gradually increase as the thoron decays and after a while become too long for convenient measurement. Graphing the inverse of the fall time as a function of time gives a plot of the exponential decay of the thoron. For a more accurate measurement, plotting directly on semi-log paper will give a straight line whose slope can be used to determine the half life.
Setting it up:
There are two windows in the electroscope. Our video cameras are sensitive enough to see the gold leaf without additional illumination. For good contrast, it is best to put white paper on the outside of the rear window. A reference line, to measure the leaf against, can be drawn on the paper or either window.
First check the background: charge the electroscope and, without introducing thoron, measure the time required for the leaf to fall to the reference line. The time should be long (200 sec or more); this "background" fall rate represents the rate at which imperfect insulation and ionization induced by cosmic radiation drains charge from the electroscope 4 and should be subtracted from the data if accuracy is important.
Next make a trial run to master the somewhat tricky technique of taking data. Here is one suggested method. After charging the electroscope, squeeze the rubber bulb gently several times until the leaf falls to the reference mark in 2 or 3 seconds. Then charge the electroscope, starting the clock as you push the charging button. Note the time at which the leaf just completes its first traversal to the mark and, without stopping the clock, recharge the electroscope. The recharging should be simultaneous with the reading of the clock. Note the time at which the leaf completes its second traversal and at just that instant, again charge the electroscope. Continue until the leaf requires over 100 seconds to fall to the mark. You will have a list of about 10 to 15 times. The difference between consecutive readings in this sequence represents the fall time.
Comments:
In addition to being an excellent demonstration of radioactive decay, the historical spirit and measurement techniques are also preserved in this presentation. The half life of thoron lends itself very nicely to the typical time frame of a lecture demonstration. There are few such radioisotopes as convenient as this. This demonstration could also be used in the context of a discussion of our present day concern over the problem of indoor radon pollution (see footnote 2 in regard to the other radon isotopes).
1 E. Rutherford, Phil Mag 49, 1, 1900. The experiment is of historical significance since the concept of matter decaying, together with Planck's quantum theory, signaled the end of the classical era of physics.
2 F.W. Walker, D.G. Miller and F. Feiner, Chart of the Nuclides, (General Electric Co., San Jose CA, 1984). A very nice historical account of its discovery by Rutherford and Soddy is given by Steven Weinberg, The Discovery of Subatomic Particles, (Scientific American Library, N.Y., 1983), pp 116-119. The other naturally occuring isotopes of radon are products of the 238U-Radium series and the 235U-Actinium series (radium emanation is 222Rn with a half life of 3.82 days and actinium emanation is 219Rn with a half life of 3.96 seconds).
3 K.T. Bainbridge and J.C. Street, Am J Phys 6, 99 (1938). "Laboratory and demonstration experiments on the law of radioactive decay and the determination of the disintegration constant of thoron"
4 Indeed, it was this discovery of the leakage of electric charge from electroscopes that led researchers to the discovery of cosmic rays.